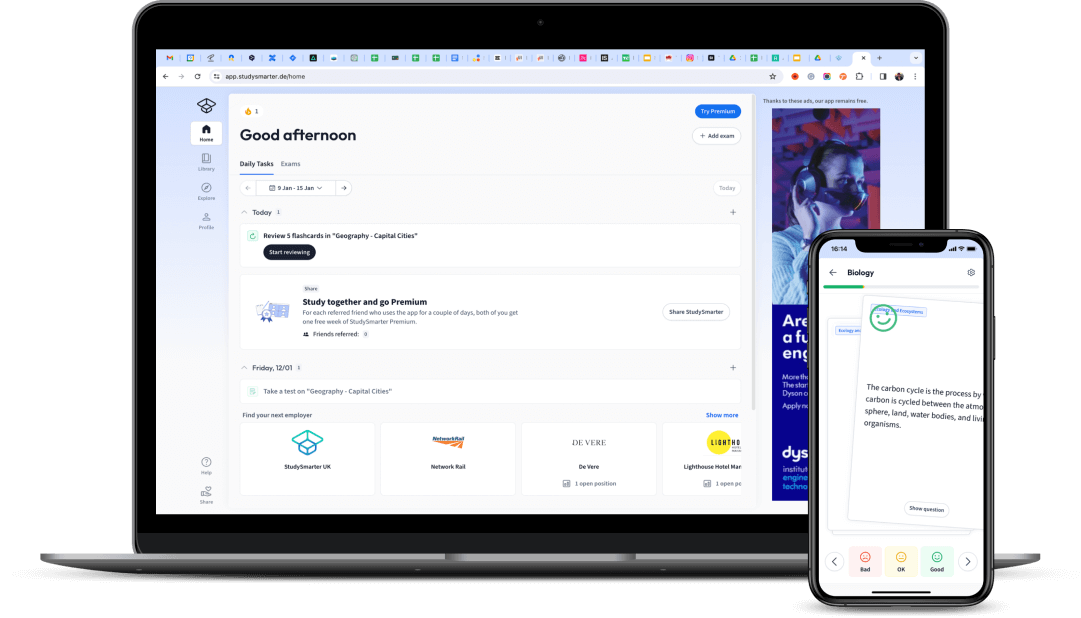
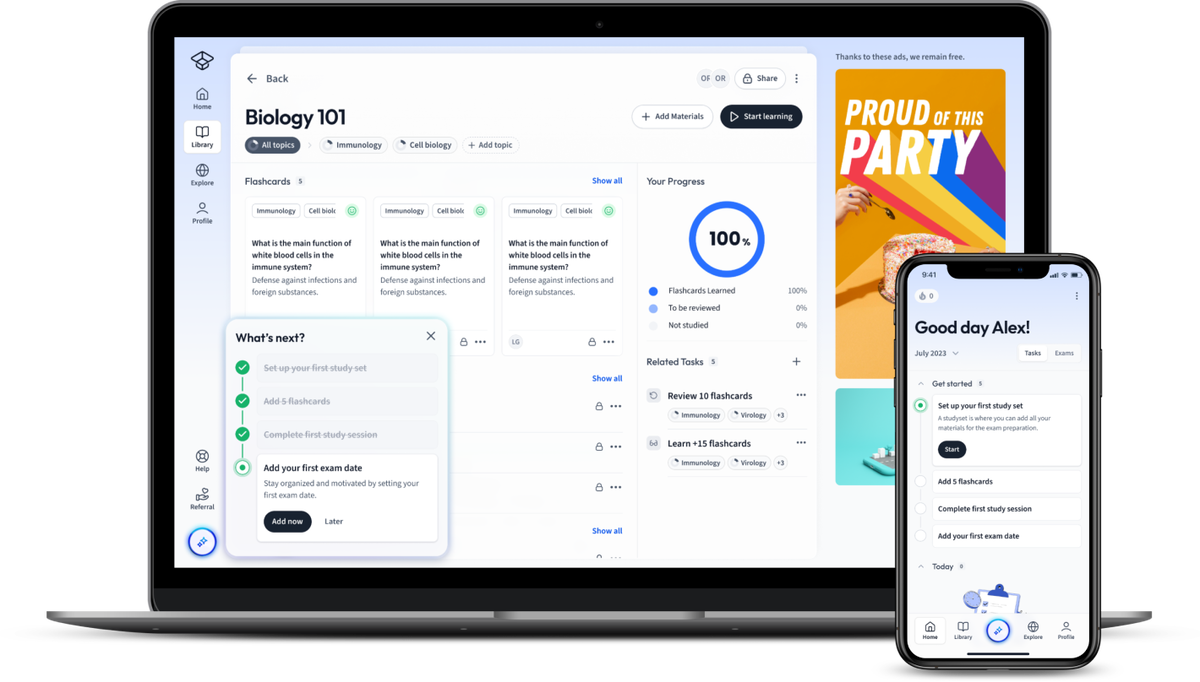
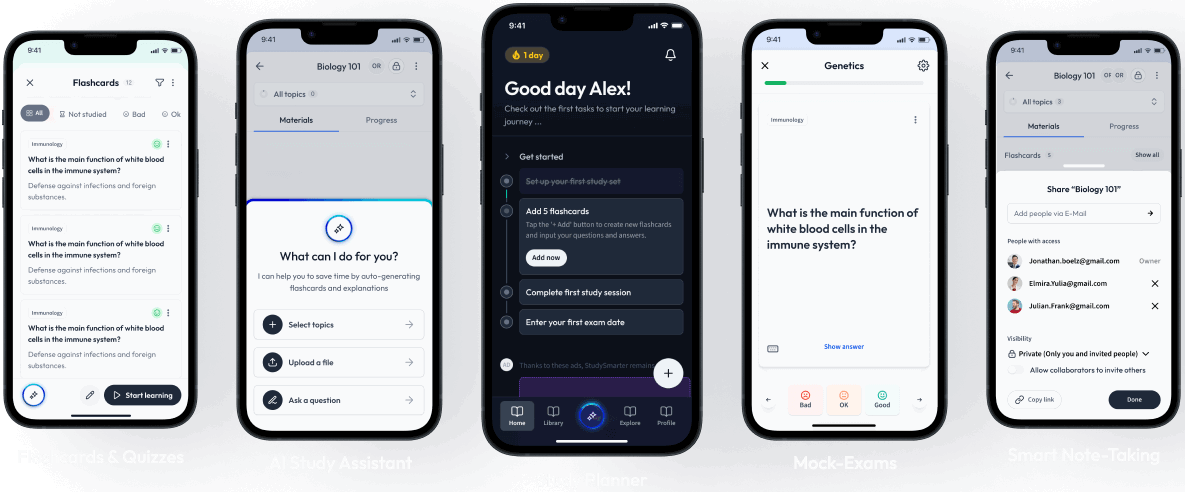
Find Study Materials for
-
Explanations
-
Exams
-
Magazine

Main menu